
CAR T cells targeting the B cell marker CD19 have been impressively effective in the treatment of B cell malignancies such as acute lymphoblastic leukemia and lymphomas, recently gaining US Food and Drug Administration (FDA) approval ( 13). Upon recognition of specific antigens on target cells, CARs trigger the release of cytokines and cytotoxic mediators. CARs consist of an extracellular antigen-binding moiety that is fused via a transmembrane region to an intracellular signaling domain derived from the T cell receptor complex and from costimulatory molecules ( 13, 14). One important application of ON-switches is the regulation of T cells that are genetically engineered to express chimeric antigen receptors (CARs). Thus, an effective molecular ON-switch that can induce heterodimerization in a clinically relevant setting is still lacking. However, their clinical translation is limited due to unfavorable characteristics of the small molecule or the nonhuman origin of protein components ( 7, 9– 12). Indeed, various systems have been introduced that enable such conditional heterodimerization ( 4– 8), including the FRB/FKBP system that is used extensively in vitro. However, for many applications it is necessary to regulate the interaction of two different proteins. The only molecular ON-switch that has been used in humans in vivo is based on a mutated version of FK506 binding protein (FKBP) 12, which is homodimerized upon administration of the small molecule AP1903 ( 3). In general, in a CID the interaction between two proteins can be triggered by a small molecule, and therefore, CID systems can also be regarded as molecular ON-switches. 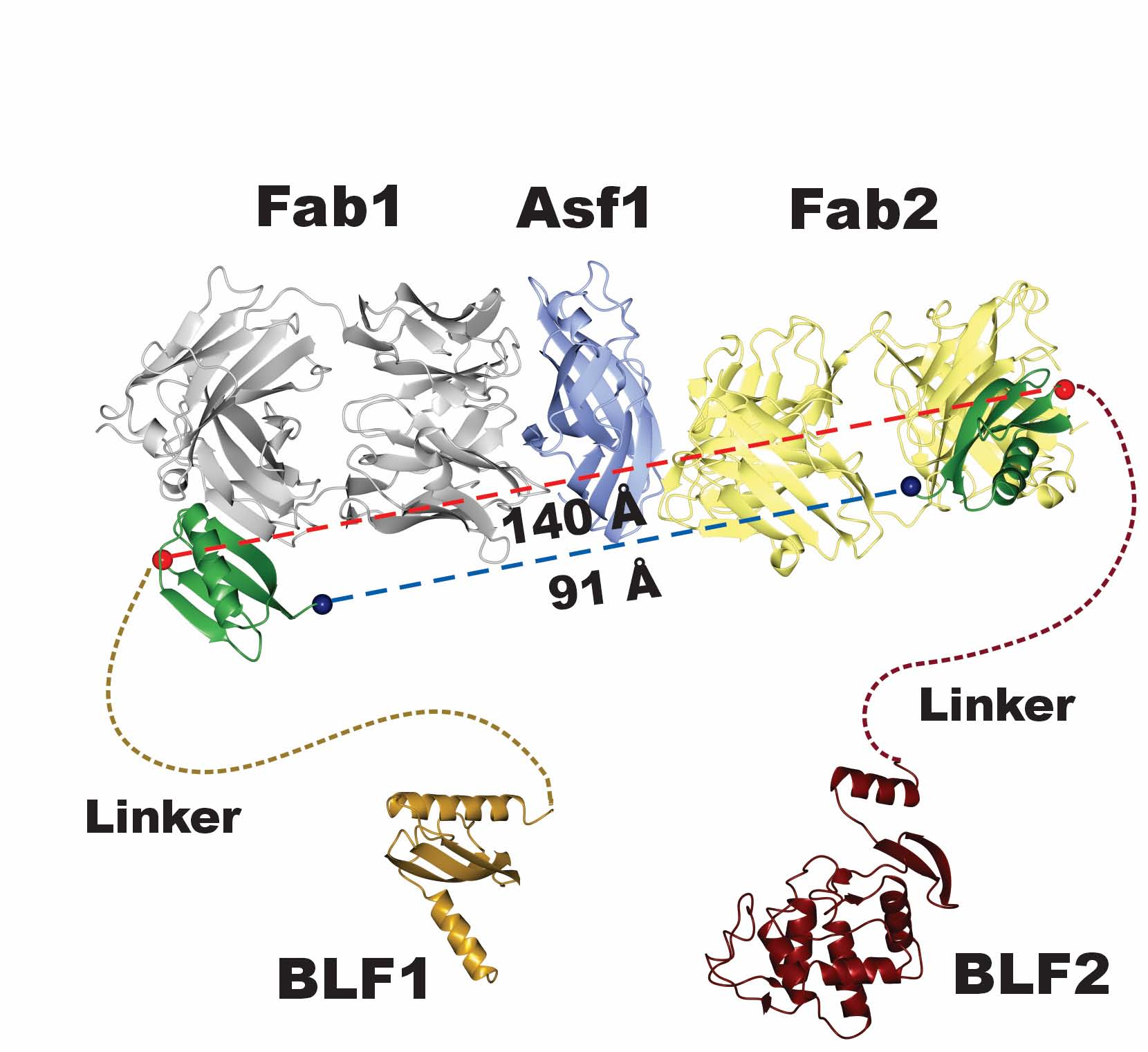
These switchable systems are commonly known as chemically induced dimerization (CID) systems ( 1, 2). The ability to control protein–protein interactions with small chemical compounds can open up exciting applications across various fields such as cell biology, immunology, and immunotherapy. We anticipate that lipocalin-based ON-switches have the potential to be broadly applied for the safe pharmacological control of cellular therapeutics. Furthermore, the ON-switch successfully regulated the activity of primary human CAR T cells in vitro. We demonstrate that this conformation-specific ON-switch is highly dependent on the presence of A1120, as demonstrated by an ∼500-fold increase in affinity upon addition of the small molecule drug. The crystal structure of an assembled ON-switch shows that the engineered binder specifically recognizes the conformational changes induced by A1120 in two loop regions of hRBP4. Two different protein scaffolds were engineered to bind to hRBP4 when loaded with the orally available small molecule A1120. Here, we developed an ON-switch system in which the human retinol binding protein 4 (hRBP4) of the lipocalin family interacts with engineered hRBP4 binders in a small molecule-dependent manner. ON-switches based on clinically applicable compounds and human proteins would greatly facilitate their therapeutic use. Molecular ON-switches in which a chemical compound induces protein–protein interactions can allow cellular function to be controlled with small molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
